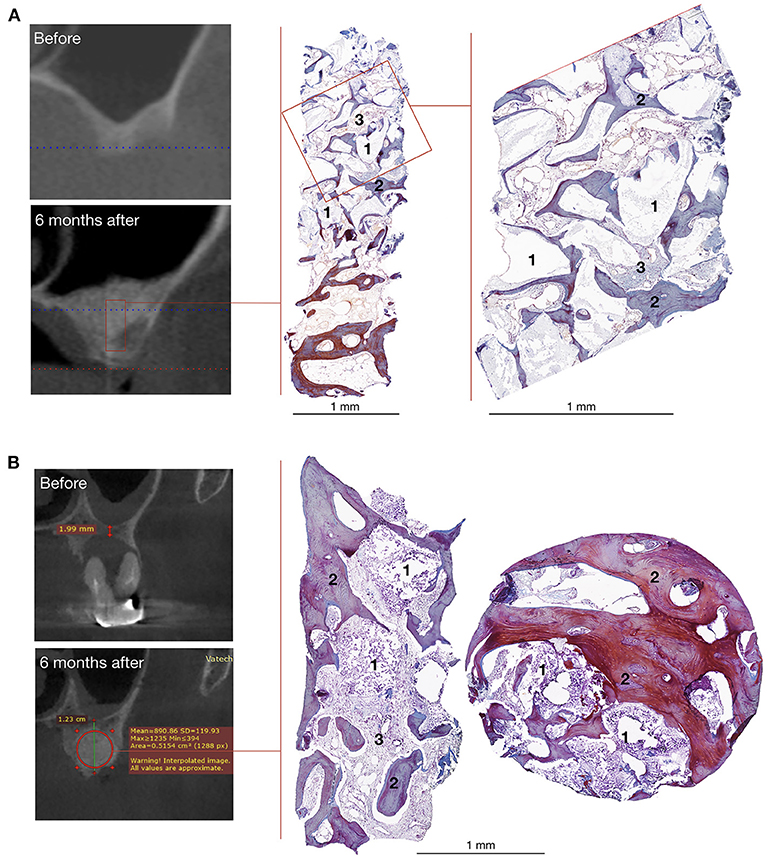  
Paul Arnold, Rick Sasso, Michael Janssen, Michael Fehlings, Robert Heary, Alex Vaccaro and Branko Kopjar.Īt 2 yr, the fusion rate was 97.30% and 94.44% in i-FACTOR and autograft subjects, respectively(P=.2513), and neurological success rate was 94.87%(i-Factor) and 93.79% (autograft P=.7869). The clinical data was published in Neurosurgery in September 2018. P-15 is a novel synthetic, 15-amino-acid polypeptide that, according to the referenced study, "mimics the cell-binding domain of type I collagen and is able to signal a mechanical and biochemical communication pathway that ultimately results in new bone formation." It is in a new category of bone graft technology and is one of only two drug-device combination products approved by the FDA." The drug-device combination, according to the company, is based on synthetic small peptide (P-15) technology "that accelerates new bone formation in patients with degenerative disc disease. and around the world.”Ĭerapedics commercialized the bone graft in 2016. He continued that he believes i-FACTOR Bone Graft "represents one of the most important technological breakthroughs in this field and are committed to compiling extensive Level I human clinical data to support its use across the U.S. said the approval allows labeling to reflect "longer-term clinical data showing that the statistical superiority to autologous bone in overall clinical success that was observed at one year have been maintained at two-year follow up." 
On November 18, 2019, Cerapedics announced the change to i-FACTOR’s Premarket Approval (PMA) supplement for ACDF.Ĭompany President and CEO Jeff Marx, Ph.D. asked the FDA if they could update their package insert accordingly. Peptide enhanced bone graft (e.g.New, two-year follow-up data for i-FACTOR™ Peptide Enhanced Bone Graft shows that the one-year superiority over autologous bone grafts in anterior cervical discectomy and fusion (ACDF) procedures for degenerative cervical disc disease, is sustained for two years.Demineralized bone matrices (DBM) regulated as medical devices by the FDA (e.g., Grafton Putty/Gel (Medtronic), DBX (MTF/Depuy Synthes), Accell Evo3 (IsoTis), and Propel Putty/Gel (NuVasive)).Synthetic bone graft extenders (e.g., Formagraft (NuVasive), Mastergraft (Medtronic), Vitoss (Stryker), Actifuse (Baxter), nanOss (RTI Surgical), Fibergraft (Prosidyan/Depuy Synthes), and ChronOs (Depuy Synthes)).Bone morphogenic protein (BMP) (i.e., Infuse (Medtronic)).Use of any bone graft that is not cleared by the applicable regulatory body for use within interbody implants. Understands the conditions of enrollment, is willing to follow medical advice including postoperative activity restrictions per the surgeon's standard of care, and is willing to sign an informed consent form to participate in the study.Able to undergo surgery based on physical exam, medical history, and surgeon judgment.With interbody fusion using autograft and/or allograft (i.e., cancellous and/or corticocancellous allograft bone) and NuVasive supplemental internal fixation cleared by the applicable regulatory body for use in the cervical spine.Using one of the following implants (NuVasive, Inc., San Diego, CA): Have a planned spine surgery using interbody implants at one or more cervical levels (C2-T1) for degenerative disc disease and/or cervical spinal instability.Patients who are ≥18 years of age at the time of consent.Why Should I Register and Submit Results?.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
